|
|
|
|
News The Project Technology RoboSpatium Contribute Subject index Download Responses Games Gadgets Contact <<< Observational error Current, voltage >>> TemperatureTerminologyTemperature is a physical property which quantitatively expresses the common notions of hot (=high temperature) and cold (=low temperature). The temperature of an object is scale invariant, meaning it does not depend on the size of the object, hence temperature is an intensive property.Macroscopic objects are composed of many particles, such as atoms or molecules. On the atomic level, temperature is the result of the motion of the particles the object consists of. Each moving or rotating particle carries kinetic respectively rotational energy according to it's mass, shape and speed. If an atom is fixed in a crystal lattice, the potential energy resulting from the electric fields has to be considered, too. In a statistical mechanics approach, temperature increases as this motion and thus the kinetic energy increases. Temperature is measured with thermometers, which are calibrated to the Kelvin scale, according to the International System of Units. The null point of the Kelvin scale is the absolute zero, the temperature at which all thermal motion of atoms stops. This is the first reference point of the scale. The second one is the triple point of water. When heating up water, it starts boiling at a certain temperature AND pressure. At those condition, liquid water and water vapor can coexist in a stable equilibrium. The density of water vapor is significantly lower than those of liquid water, hence water vapor is transformed into liquid water while the pressure increases at constant temperature. Another phase change occurs while solid water (ice) is melting at increasing temperatures. The density of ice is lower than those of liquid water, hence water is freezing and turned into ice if the pressure decreases at constant temperature. There is a single combination of pressure and temperature, at which liquid water, solid ice, and water vapor can coexist in a stable equilibrium. The pressure at the triple point of water is 611.73 pascals. The value of the Kelvin scale at those second reference point is set to exactly 273.16 K. One kelvin is defined as 1/273.16 of the difference between these two reference points. Those "curious" value was chosen to match the magnitude of the very common Celsius scale. So 0K is exactly -273.15°C. Liquid-in-glass thermometer Figure 1:
Figure 1:This a very common type of thermometer and the functionality is based on the expansion of a liquid at increasing temperatures. Most of the liquid is stored inside of the bulb at the bottom of a narrow glass tube and with increasing temperature the length of the liquid inside the tube is increasing, too. Calibrated marks on the tube allow the temperature to be read. The whole system is closed to avoid the vaporisation of the liquid and the space above the liquid is often filled with nitrogen gas with less than the atmospheric pressure. Mercury is the most common liquid inside of this kind of thermometer. Liquid-in-glass thermometers are qualified to detect the ambient temperature. Several disadvantages have to be considered while using this type of thermometer: 1.) Depending on the amount of liquid inside of the bulb it takes a long time until the whole volume has reached its state of equilibrium. Quick changes in temperature can't be detected. 2.) The whole thermometer or at least the whole bulb must be heated up, to be able to measure the temperature. It is impossible to determine the temperature of a solid body whose temperature differs from the ambient temperature. 3.) The temperature range of a mercury-in-glass thermometer can't fall below the melting point (-38.83 °C) or rise above the boiling point (356.73 °C). When using other liquids, the temperature range can be extended. Bimetallic strip thermometer Figure 2:
Figure 2:Different materials expand at different rates while they are heated. If two metals are joined together throughout their length by soldering or welding, the different expansions force the flat strip to bend one way while heating up and in the opposite way while cooling down. While heating up, the metal with the higher coefficient of thermal expansion is on the outer side and it is at the inner side while cooling below its initial temperature. An indicating needle attached to one end of a bimetallic coil operates like a thermometer. The disadvantages of bimetallic strip thermometers are similar to those of the Liquid-in-glass thermometers, because the whole coil has to be heated up to indicate the temperature. You can determine the current temperature of the rover universe with the help of the thermometer displayed at the photo. Resistance Thermometer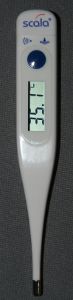 Figure 3:
Figure 3:The conductivity of some materials varies significantly with temperature. Resistance thermometers are detecting the electrical resistance of the sensor which is why they require a power source to operate. A common material is platinum, because of its linear resistance-temperature relationship and its chemical inertness. Carbon resistors are inexpensive and very reliable at extremely low temperatures. Thermo sensors like that are very compact which is why they heat up or cool down quickly. They can also detect the surface temperature of solid objects. Thermocouple Figure 4:
Figure 4:Even smaller than the sensors of resistance thermometers, are thermocouples which simply consist of a junction between different metal wires. Related to the temperature difference between the temperature at the junction and those at the ends of the wires, a voltage is generated. Thermo sensors based on this principle are inexpensive and can measure a wide range of temperatures. A disadvantage is the accuracy. Temperature differences below one degree Celsius are difficult to achieve. <<< Observational error Current, voltage >>> News The Project Technology RoboSpatium Contribute Subject index Archives Download Responses Games Links Gadgets Contact Imprint |
|
|